Balloon
Background
A balloon is an air-tight bag made out of a light material that can be inflated with air or gas. Toy balloons are available in all kinds of shapes, sizes, and colors to delight children and adults at birthday parties and other festive occasions.
Balloons were first invented in France in the late 18th century. Two papermakers, Jacques and Joseph Montgolfier, discovered that when paper bags are filled with hot air, the bags rise. Quick to realize the potential of this, they began experimenting with balloons of various materials such as paper, cloth, and silk. They made the first public demonstration of a lighter-than-air balloon in June 1783, with a 35-foot (11 m) diameter balloon made of cloth lined with paper. Later that year, Jacques Charles flew a balloon made of silk coated with a rubber varnish and filled with hydrogen, a gas that is lighter than air. These early demonstrations attracted a great deal of excitement, and balloons were soon put to many uses in science, sport, and war.
The rubber toy balloon as we know it today is different from the early balloons in that it is made entirely of rubber. A practical way of making such formed rubber products required several discoveries and inventions. These developments took place gradually over the years since the first rubber factory in the world was established near Paris in 1803.
Natural latex is a mixture of small globules of rubber substance suspended in water (much like milk). When it is exposed to air, heat, or certain chemicals, it coagulates or clots together. The globules of rubber lump together and separate from the watery portion of the latex, eventually forming an elastic, solid material. To improve its strength, resilience, and resistance to hot and cold temperatures, rubber is vulcanized or cured by various methods, such as mixing with certain chemicals or treating with heat.
The idea of making a product out of rubber is an old one. The natives of South America created bottles and other articles by coating molds made of earth long before Europeans began experimenting with rubber in the mid-1700s. In 1830, the Englishman Thomas Hancock patented a process for creating products by pouring latex over molds or dipping molds into a latex mixture—the forerunner of the modern technique of producing dipped products such as rubber gloves and condoms.
In 1921, a method of retarding the coagulation of liquid latex was developed. This method enabled rubber makers to transport raw latex in a liquid form more easily to manufacturing centers around the world. This in turn led to new processes for making rubber goods. In the early 1920s, a number of patents were granted in England for processes that allowed molds to be dipped in liquid latex. In 1931, the first modern latex balloon was created by Neil Tillotson in his attic. He sold 15 of his "Tilly Cat" balloons (shaped like a cat's head, complete with whiskers printed on with dye) for the Patriot's Day parade in Massachusetts in April 1931, and formed a company that still makes balloons today.
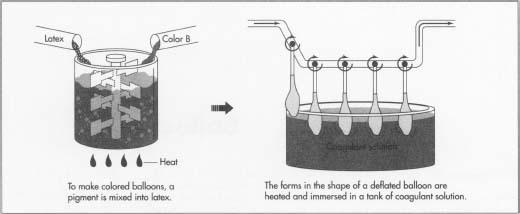
Raw Materials
Although rubber can be made synthetically, natural latex is preferred for its great elasticity. It can be stretched to seven or eight times its original length and still return to its former shape. Synthetic rubber has not proven to be as elastic and resilient as natural latex.
Raw, natural latex is a white or yellowish opaque liquid, similar in appearance to milk. Latex is the secretion of certain plants, in particular the Hevea tree originally found in Brazil. The most important sources of natural rubber today are plantations in Malaysia and Africa.
Producers of rubber must harvest the raw material from these trees, which involves scoring the trees with shallow cuts and letting the sap ooze from the cuts into buckets. The latex is collected in large containers, filtered to remove foreign particles, and mixed with alkali to prevent coagulation. It is then shipped in liquid form to processing centers in different parts of the world.
Latex must be mixed with additives before it can be used in industrial processes. Certain chemicals are mixed in to achieve a desired thickness, rate of drying, and other properties. Other chemicals (collectively known as antidegradants) are added to slow the oxidation and decomposition of the rubber. To give it color, pigments are mixed into the latex. The pigments may be fine metal oxide powders or organic dyes.
The Manufacturing
Process
In essence, the process of making a toy balloon involves dipping a mold into liquid latex. The mold, or form, is shaped like a deflated balloon.
The earliest balloon forms were disposable, made from cardboard attached to dowels. Modern forms are reusable and usually made from stainless steel, aluminum, or porcelain. The forms must be smooth and polished. A number of such forms are attached upside down to a board or rack. The boards are moved mechanically from one station to another in the factory.
To be efficient in terms of cost and number of balloons produced, balloon manufacture has become a highly automated, continuous loop process. Balloons are made in batches, all of the same color and size, since changing the color and form is time-consuming and requires manual intervention. Manual intervention is usually only needed for setting up a run and then later for packaging the finished product, and for dealing with occasional mechanical problems that may arise.
Preparing the latex
-
1 Prior to its use, the latex may need to be colored. This involves
mixing a pigment into the latex. It may be done at the balloon factory,
or the balloon maker may purchase already-pigmented latex from a
supplier.
- 2 The latex must be poured into tanks into which the forms will be dipped. The tanks are kept at a certain temperature and may include stirring mechanisms to keep the latex circulating to avoid settling.
Dipping the forms
- 3 The balloon forms are first heated, then immersed in a tank of coagulant solution for a few seconds. When the forms are immersed in the liquid latex, the coagulant will cause the rubber to gel in a thin sheet around the forms. A commonly used coagulant solution is a mixture of water, a calcium-based salt, soap, and talc powder. The salt is the actual coagulant; the soap helps the latex spread in an even film, and the talc helps ease the removal of the rubber from the forms in a later step.
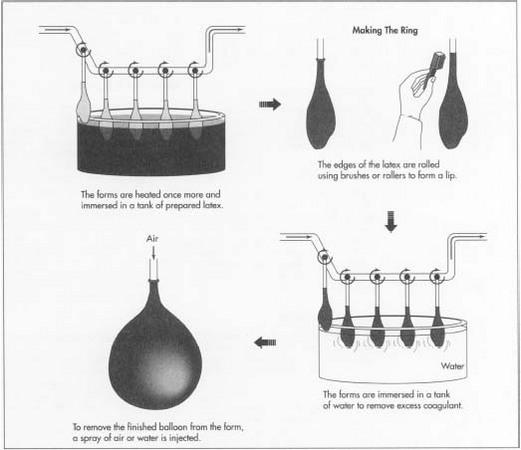
- 4 The forms are heated to a temperature between 100°F (38°C) and 200°F (93°C), and then immersed in a tank of colored latex. The coagulant causes the latex to coat the forms. The longer the forms are left in the tank, the thicker the coating that sticks to them. For balloons, a very thin layer of latex is desired, so the forms are immersed only for a few seconds. The forms must be inserted and removed at carefully controlled speeds to avoid trapping air bubbles and to achieve an even, thin coating.
Making the ring
- 5 A lip is formed on the neck of the balloon by rolling the edges of the rubber using brushes or rollers. This creates the ring seen around the opening of the balloon.
Removing excess coagulant
- 6 Next, the forms are immersed in a tank of leaching solution (often plain water) to dissolve and leach away excess coagulant from the rubber.
Curing the rubber
- 7 The rubber on the forms must be dried and cured. The method used varies among manufacturers. Some balloon makers use a latex that already contains a vulcanizing agent, in which case the rubber is dried at a moderate temperature. Other makers induce vulcanization by putting the rubber-coated forms into an oven and curing for as long as an hour.
Removing the balloons
- 8 The balloons are then mechanically removed from their forms. One approach is to blow them off using a spray of water or air and collecting the balloons in a basket or net.
- 9 If the balloons are removed using a spray of water, they are next placed in a centrifuge, where excess water is removed by spinning the balloons around at high speed.
- 10 The balloons are then dried in large tumble dryers.
Printing and packaging
- 11 Next, the balloons may either be packaged, or first printed and then packaged. If they are packaged directly, they are moved on a conveyor belt past a counting device and placed into bags. When an appropriate number of balloons has been placed in each bag, the bags are sealed.
- 12 Printing designs on balloons, such as logos or faces, actually involves several steps. First, the balloons must be inflated in order to allow even printing. This requires a worker to manually place each balloon on the inflating device. Next, a pattern is carefully printed on each balloon. Finally, the balloons are removed and passed on to the packaging stage.
Quality Control
The balloon manufacturing environment must be strictly controlled in order to achieve high quality and consistency. Throughout the manufacturing process, computer-based instrumentation records and controls air humidity, air temperature, latex tank temperature, the temperature in the ovens, dryers, and other parameters.
The latex and other chemicals used in the process must be carefully formulated for specific properties, and carefully maintained. For example, the latex must have certain viscosity and speed of drying. The tanks in which it is held must have devices to keep the latex circulating to avoid forming a "skin," and to prevent ingredients from settling.
Byproducts/Waste
It is in the manufacturers' best interests to waste as little rubber as possible because the cost of latex is high compared to the selling price of individual balloons. Balloon makers also reclaim much of the coagulant that ends up in the leaching solution. Unfortunately, what is not reclaimed ends up as liquid waste in the environment. The amount of chemical waste that can be released by a factory is regulated by government laws. Balloons also result in some waste after they are manufactured because they are invariably thrown away after they deflate or pop. However, because latex is natural, it eventually breaks down into other substances.
Safety Concerns
Toy balloons can be a source of joy, but they can also be unexpectedly hazardous. Young children have been known to die from accidentally choking on balloons. Latex balloons may also end up in water, where they eventually lose their color and can resemble jellyfish. Sea animals such as whales and turtles have attempted to eat them and have died because the latex clogs their digestive systems.
The Future
The toy balloon industry is very competitive. Manufacturers are constantly looking for ways to make the process more automatic and efficient, especially by reducing manual intervention. Currently the most labor-intensive portions are the printing and packaging steps. Increasing automation in these steps is an area for potential future improvement.
In recent years balloons made of metal films have become popular. The manufacturing process of these balloons is very different. They are made from a sandwich of two swatches of mylar—a polyester film—often circular in shape, which are sealed together around the edges. A small opening is left through which the balloon may be inflated. Because the material is initially flat, these balloons can be printed more easily than balloons made of rubber. The foil can be made very shiny and reflective, allowing for very bright designs. They are stronger and more durable than rubber balloons, but for some uses, this is also a disadvantage. For example, they cannot be twisted into various shapes nor can they be filled with water. The foil also takes much longer to degrade in the environment than rubber.
Where To Learn More
Books
Barlow, Fred W. Rubber Compounding: Principles, Materials, and Techniques. Marcel Dekker, Inc. 1988.
Coates, Austin. The Commerce in Rubber: The First 250 Years. Oxford University Press, 1987.
Hofmann, Werner. Rubber Technology Handbook. Oxford University Press, 1989.
— Renee M. Rottner