Shellac
Background
Lac is the name given to the resinous secretion of the tiny lac insect (Laccifer lacca) which is parasitic on certain trees in Asia, particularly India and Thailand. This insect secretion is cultivated and refined because of the commercial value of the finished product known as shellac. The term shellac is derived from shell-lac (the word for the refined lac in flake form), but has come to refer to all refined lac whether in dry or suspended in an alcohol-based solvent.
Shellac is primarily used as a wood sealer and finisher today. It has the great advantage of being soluble in ethyl or denatured alcohol, an environmentally-safe solvent. Alcohol solvents also render shellac a quick dry—shellac coatings on wood generally dry in about 45 minutes, as opposed to oil finishes which take many hours to dry. In addition, shellac does not fade in sunlight or oxidize over time. However, shellac has a limited shelf life and may not dry properly if it has exceeded the shelf life recommended by the manufacturer. This shelf life may be as short as six months or as long as three years depending on the manufacturer's additives.
Industrial uses for shellac include floor polishes, inks, grinding wheels, electrical insulations, and leather dressings. This natural, resinous sealer is non-toxic and is Federal Drug Administration (FDA) approved for use to coat candies, pharmaceuticals, fruit, and baby and children's furniture.
Shellac is available at most hardware or paint stores in clear or white shellac or orange shellac, which imparts an orange-red tint to natural wood. Other tints derive their color not from dyes or bleaches, but because of the tree to which the lac bug has attached itself—the sap affects the color of the bug secretions thus altering the color of the refined shellac. Shellac may be applied to wood, over varnish, paint, glass, ceramics, even plastic with remarkable adherence, but it cannot be used under synthetic sealers such as polyurethane.
History
Lac has been cultivated for three centuries. For most of that time, the lac bug secretions were valued for the purple-red dye derived from being soaked in water. This dye was used to color silk, leather, and cosmetics and was cultivated primarily for this purpose until the 1870s. Then aniline or chemical dyes began to supplant these and other natural dyes.
As early as the sixteenth century, references were made to the usefulness of the lac bug secretions as a decorative lacquer for furniture and fine musical instruments. Natives of the Far East had laboriously cultivated and processed the shellac by hand, scraping the branches encrusted with the lac bug secretions, forcing the secretions into muslin, and holding long muslin bags of the secretions over the fire to liquefy and purify it. They pulled it by hand into huge sheets and then broke the sheets into flakes for re-moisturizing later.
Hand processes were partially replaced by the mid-nineteenth century. Just as the lacderived dye was about to fade in popularity, industrial plants began processing the lac secretions for use as a wood sealer and finish. In 1849, William Zinsser founded Wm. Zinsser & Company in New York. Zinsser's shellacs were soluble in ethyl alcohol and were the first quick-drying, tough, colorless
Raw Materials
Shellac is generally made from two ingredients, raw seed lac and ethyl alcohol. In fact, most companies want to purify shellac as completely as possible—impurities from the bug, the cocoon etc. are removed, as are natural waxes. Shellac is generally shipped in dry or flaked form and is re-moisturized with an alcohol solvent, generally denatured alcohol. Some companies add ingredients to lengthen the shelf life of their product but will not reveal these proprietary additives. Shellac that is bleached (or made into clear shellac) are dissolved in sodium carbonate and centrifuge to remove insolubles and then bleached with sodium hypochlorite.
The Manufacturing
Process
The role of the lac bug
-
1 Shellac is produced by a tiny red insect. Swarms of the insects feed
on certain
trees, primarily in India and Thailand, known informally as lac trees. The lac bugs' life cycle is only six months, in which time they eat, propagate, and secrete the resin they've taken in from the tree to produce shellac.
In certain seasons of the year, these insects swarm in huge numbers on the trees, settle on branches, and project protrusions into the tree to penetrate the bark. They suck up the sap and absorb it until they feed themselves to death (called the feast of death amongst the indigenous peoples). At this same time, propagation continues, with each female lac bug laying about 1,000 eggs before dying.
The sap is chemically altered in the lac bug's body and is then exuded onto the tree branch. On contact with the air, the excretion forms a hard shell-like covering over the entire swarm. This covering forms a crust over the twig and insects. As the female lac bug is exuding the ingested sap she is preparing to die and is providing a fluid in which her eggs will mature under protection. The males' role is to fertilize the female, and it is after fertilization that the females' lac output is vastly increased. The adult males and females become inactive, and the young start to break through the crust and swarm out.
Refining the crusty resin
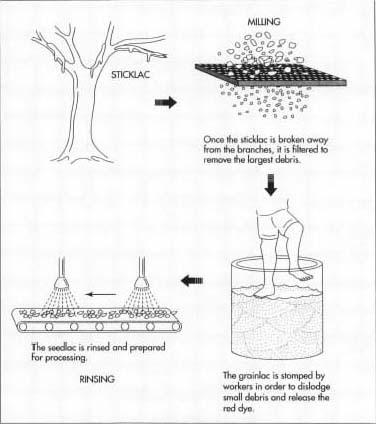
- 2 Workers cut millions of encrusted branches, called sticklac, for transportation to refineries of some sort (either handrefined or mechanically refined). Some workers use mallets and break off the crusty coating much as ice is broken from branches in the winter (it is referred to as grainlac).
- 3 At refining centers, sticklac is scraped to remove the secretions from the twigs. Sticklac and grainlac is ground with rotating millstones. The resulting ground material is quite impure, containing resin, insect remains, twigs, leaves, etc. The mixture is forced through a screen, removing the largest of the impurities.
-
4 The sifted resin mixture is put into large jars and stomped by a
worker to crush granules and force the red dye from the lac seeds and
the insect remains will be freed from the resin. Dye water, scum, and
other impurities are then washed away in several rinsings. The mixture
is spread out on a concrete floor to dry and called seedlac because it
resembles seed. Seedlac is the raw material from which both orange
shellac and bleached or clear shellac are produced.
Shellac may be made from seedlac by hand or by modern mechanical equipment. Nearly all American-used shellac is refined with the help of machinery, using a heat-or solvent-based process.
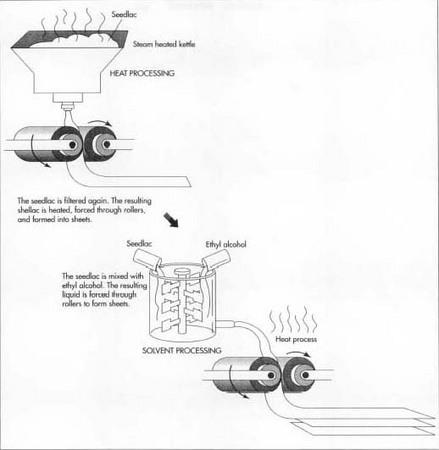
Heat process
- 5 Seedlac is melted onto steam-heated grids. The molten lac is forced by hydraulic pressure through a sieve or screen, either of cloth or fine mesh. The filtered shellac is collected and transferred to a steam-heated kettle, which then drops the molten liquid onto rollers. The liquid is squeezed through the rollers and forced into large, thin sheets of shellac. When dry, this shellac sheet is broken into flakes and transported to another area in which the flakes are combined with denatured alcohol to produce the consumer's shellac.
Solvent process
- 6 In this process, the seedlac and solvent, usually ethyl alcohol, are mixed in a dissolving tank, refluxed for about an hour and then filtered to remove impurities. The filtered resin is sent through evaporators that remove the alcohol solvent, rendering it a viscous liquid. This liquid is then dropped onto rollers, which force it into sheets. The sheets are then are dried and flaked apart.
Bleached shellac
Despite the removal of much of the red dye from the lac seeds in the refining process, shellac remains an orangish solution after processing is complete. Some consumers prefer a clear shellac finish, so manufacturers have developed a way to bleach the color from the shellac.
- 7 Bleaching begins with dissolving seedlac, which is alkali-soluble, in an aqueous solution of sodium carbonate. The solution is then passed through a fine screen to remove insoluble lac, dirt, twigs, etc. The resin is then bleached with a dilute solution of sodium hypochlorite to the desired color. The shellac is then precipitated from the solution by the addition of dilute sulfuric acid, filtered, and washed with water. It is dried in vacuum driers and ground into a white powder ready for shipment to a plant that will add liquid to the flakes.
Mixing shellac for the consumer
-
8 Large shellac manufacturers are shipped the dry shellac flakes. They
then remoisturize the flakes by adding denatured ethyl alcohol. Shellac
is offered to the consumer in flake form or suspended in denatured
alcohol. It is the latter than is most popular with the consumer.
Manufacturers of shellac refer to the concentration of shellac flakes to
denatured alcohol in terms of pounds of cuts—the number of pounds
of
shellac flakes dissolved into a single gallon of denatured alcohol.
Thus, a one pound cut of shellac contains one pound of shellac flakes
dissolved in a gallon of alcohol—very dilute shellac. The
manufacturers' standard cut offered to the consumer pre-mixed is
termed a three pound cut. Some consumers then dilute it further with
denatured alcohol if they so desire.
The most popular shade of shellac sold premixed is the orange shellac although clear or white shellac is also offered pre-mixed to the consumer. Manufacturers always stamp the date of mixing of the shellac into the can. Each manufacturer has a recommended shelf life for the product and the consumer should heed that the product is not used after the period suggested by the manufacturer. If used after the time span recommended, the shellac may never dry completely.
For woodworkers who prefer the deep rich colors of garnet shellac or buttonlac, the dried flakes of these shellacs may be purchased from the manufacturer and mixed with denatured alcohol by the consumer.
Byproducts/Waste
The denatured ethyl alcohol used in the process of manufacturing shellac is a strictly regulated byproduct and is known as a volatile organic substance (VOC). The most dangerous or hazardous part, perhaps the most polluting, are the insolubles that are refined out of the sticklac and grainlac such as twigs, cocoons, leaves, bug bodies, etc. saturated with alcohol. The shellac industry is working on building huge evaporators, which will suck all the alcohol out of these insolubles so the volatility will not be an issue. Shellac flakes are all natural and non-toxic. It is the alcohol solvents that are regulated.
Quality Control
Chemical analysis does not assist in determining the quality of shellac. More important are empirical tests such as flow and shelf life that most customers have articulated as of great concern. In addition, carefully examining the purity of the shellac by removing as many of the natural impurities found within the sticklac is of utmost importance (insolubles are defined by the undissolved matter remaining when the resinous compound is mixed with hot alcohol). All refining processes are monitored for their effectiveness in removing these undesirables.
Where to Learn More
Books
Russel, M. Shellac. London: Ann Eccles and Son Ltd for Angelo Shellac, 1965.
The Story of Shellac. Somerset, NJ: Wm. Zinsser & Co., 1989.
— Nancy EV Bryk